Food
Industry with Protease
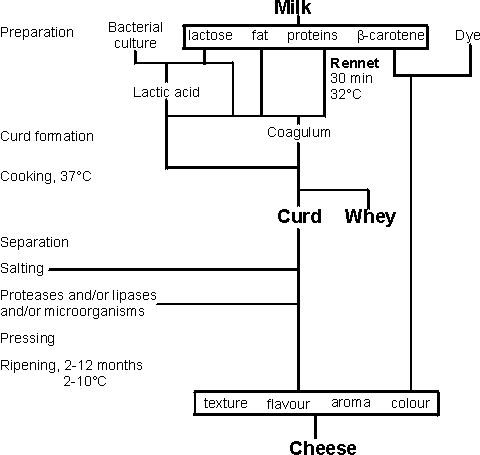
The action of rennet in cheese making is an
example of the hydrolysis of a specific peptide linkage, between phenylalanine
and methionine residues (-Phe105-Met106-) in the k-casein protein present in
milk .The k-casein acts by stabilising the colloidal nature of the milk, its
hydrophobic N-terminal region associating with the lipophilic regions of the
otherwise insoluble a- and b-casein molecules, whilst its negatively charged
C-terminal region associates with the water and prevents the casein micelles
from growing too large. Hydrolysis of the labile peptide linkage between these
two domains, resulting in the release of a hydrophilic glycosylated and
phosphorylated oligopeptide (caseino macropeptide) and the hydrophobic para-k-casein,
removes this protective effect, allowing coagulation of the milk to form curds,
which are then compressed and turned into cheese (Figure 1). The coagulation
process depends upon the presence of Ca2+ and is very temperature dependent (Q10
= 11) and so can be controlled easily. Calf rennet, consisting of mainly
chymosin with a small but variable proportion of pepsin, is a relatively
expensive enzyme and various attempts have been made to find cheaper
alternatives from microbial sources These have ultimately proved to be
successful and microbial rennets are used for about 70% of US cheese and 33% of
cheese production world-wide.

Figure 1. Outline method for the preparation of
cheese.
The major problem that had to be overcome in
the development of the microbial rennets was temperature lability. Chymosin is a
relatively unstable enzyme and once it has done its major job, little activity
remains. However, the enzyme from Mucor miehei retains activity during the
maturation stages of cheese-making and produces bitter off-flavours. Treatment
of the enzyme with oxidising agents (e.g. H2O2, peracids), which convert
methionine residues to their sulfoxides, reduces its thermostability by about
10°C and renders it more comparable with calf rennet. This is a rare example of
enzyme technology being used to destabilise an enzyme Attempts have been made to
clone chymosin into Escherichia coli and Saccharomyces cerevisiae but, so far,
the enzyme has been secreted in an active form only from the latter.
The development of unwanted bitterness in
ripening cheese is an example of the role of proteases in flavour production in
foodstuffs. The action of endogenous proteases in meat after slaughter is
complex but 'hanging' meat allows flavour to develop, in addition to tenderising
it. It has been found that peptides with terminal acidic amino acid residues
give meaty, appetising flavours akin to that of monosodium glutamate.
Non-terminal hydrophobic amino acid residues in medium-sized oligopeptides give
bitter flavours, the bitterness being less intense with smaller peptides and
disappearing altogether with larger peptides. Application of this knowledge
allows the tailoring of the flavour of protein hydrolysates. The presence of
proteases during the ripening of cheeses is not totally undesirable and a
protease from Bacillus amyloliquefaciens may be used to promote flavour
production in cheddar cheese. Lipases from Mucor miehei or Aspergillus niger are
sometimes used to give stronger flavours in Italian cheeses by a modest
lipolysis, increasing the amount of free butyric acid. They are added to the
milk (30 U l-1) before the addition of the rennet.
When proteases are used to depolymerise
proteins, usually non-specifically, the extent of hydrolysis (degree of
hydrolysis) is described in DH units where:
![]()
Commercially, using enzymes such as subtilisin,
DH values of up to 30 are produced using protein preparations of 8-12% (w/w).
The enzymes are formulated so that the value of the enzyme : substrate ratio
used is 2-4% (w/w). At the high pH needed for effective use of subtilisin,
protons are released during the proteolysis and must be neutralised:
subtilisin (pH 8.5)
H2N-aa-aa-aa-aa-aa-COO-
![]() H2N-aa-aa-aa-COO-
+ H2N-aa-aa-COO- + H+
H2N-aa-aa-aa-COO-
+ H2N-aa-aa-COO- + H+
where aa is an amino acid residue.
Correctly applied proteolysis of inexpensive
materials such as soya protein can increase the range and value of their usage,
as indeed occurs naturally in the production of soy sauce. Partial hydrolysis of
soya protein, to around 3.5 DH greatly increases its 'whipping expansion',
further hydrolysis, to around 6 DH improves its emulsifying capacity. If their
flavours are correct, soya protein hydrolysates may be added to cured meats.
Hydrolysed proteins may develop properties that contribute to the elusive, but
valuable, phenomenon of 'mouth feel' in soft drinks.
Proteases are used to recover protein from
parts of animals (and fish) would otherwise go to waste after butchering. About
5% of the meat can be removed mechanically from bone. To recover this, bones are
mashed incubated at 60°C with neutral or alkaline proteases for up to 4 h. The
meat slurry produced is used in canned meats and soups.
Large quantities of blood are available but,
except in products such black puddings, they are not generally acceptable in
foodstuffs because of their colour. The protein is of a high quality
nutritionally and is de-haemed using subtilisin. Red cells are collected and
haemolysed in water. Subtilisin is added and hydrolysis is allowed to proceed
batchwise, with neutralisation of the released protons, to around 18 DH, when
the hydrophobic haem molecules precipitate. Excessive degradation is avoided to
prevent the formation of bitter peptides. The enzyme is inactivated by a brief
heat treatment at 85°C and the product is centrifuged; no residual activity
allowed into meat products. The haem-containing precipitate is recycled and the
light-brown supernatant is processed through activated carbon beads to remove
any residual haem. The purified hydrolysate, obtained in 60% yield, may be
spray-dried and is used in cured meats, sausages and luncheon meats.
Meat tenderisation by the endogenous proteases
in the muscle after slaughter is a complex process which varies with the
nutritional, physiological and even psychological (i.e. frightened or not) state
of the animal at the time of slaughter. Meat of older animals remains tough but
can be tenderised by injecting inactive papain into the jugular vein of the live
animals shortly before slaughter. Injection of the active enzyme would rapidly
kill the animal in an unacceptably painful manner so the inactive oxidised
disulfide form of the enzyme is used. On slaughter, the resultant reducing
conditions cause free thiols to accumulate in the muscle, activating the papain
and so tenderising the meat. This is a very effective process as only 2 - 5 ppm
of the inactive enzyme needs to be injected. Recently, however, it has found
disfavour as it destroys the animals heart, liver and kidneys that otherwise
could be sold and, being reasonably heat stable, its action is difficult to
control and persists into the cooking process.
Proteases are also used in the baking industry. Where appropriate, dough may be prepared more quickly if its gluten is partially hydrolysed. A heat-labile fungal protease is used so that it is inactivated early in the subsequent baking. Weak-gluten flour is required for biscuits in order that the dough can be spread thinly and retain decorative impressions. In the past this has been obtained from European domestic wheat but this is being replaced by high-gluten varieties of wheat. The gluten in the flour derived from these must be extensively degraded if such flour is to be used efficiently for making biscuits or for preventing shrinkage of commercial pie pastry away from their aluminium dishes.